Engineering Biology to Fight Cancer
Solid tumors are heterogeneous with an immunosuppressive tumor microenvironment, but that is not slowing the search to treat the most difficult, recalcitrant cases
By MaryAnn Labant and John Schiel, PhD, April 1, 2026
Foundational preclinical data generated through research, hypothesis development, testing, and reiteration leads to the development of new approaches to treat the most recalcitrant solid tumors. As today’s next-generation therapies make their progression through clinical trials and market acceptance, another invaluable feedback loop of patient data ensues to impact even more generational advances.
Cancer—especially solid tumors—is a heterogeneous disease with many obstacles that thwart treatment, including the tumor microenvironment and its immunosuppressive effects. No one therapy is ideal for every cancer patient or indication, despite stratification attempts. And, although meaningful, personalized approaches are simply not economically feasible as a broad approach.
So, scientists do what they do best. They innovate and build on accumulated knowledge, including leveraging existing technologies in new combinations, to develop new ideas that address unmet needs. New approaches look at cellular biology and immunology for their inspiration and build on the power of existing technology and therapies. These generational advances are another step toward not only remission but also potential cures.
…
Discovering new targets
Despite the emphasis on personalized treatments based on specific driver mutations, significant overlap exists in how cancers function. “Cancer may start from different mutations, but it must accomplish the same biological objectives. That convergence creates shared vulnerabilities,” said Jon Moore, PhD, CSO at Epitopea.
These changes frequently involve epigenetic regulation, alternative splicing, and broader shifts in the proteome. As a result, conservation of certain aberrantly expressed tumor-specific antigens (aeTSAs) occurs across patients with the same tumor type—even if those proteins themselves are not classical drivers.
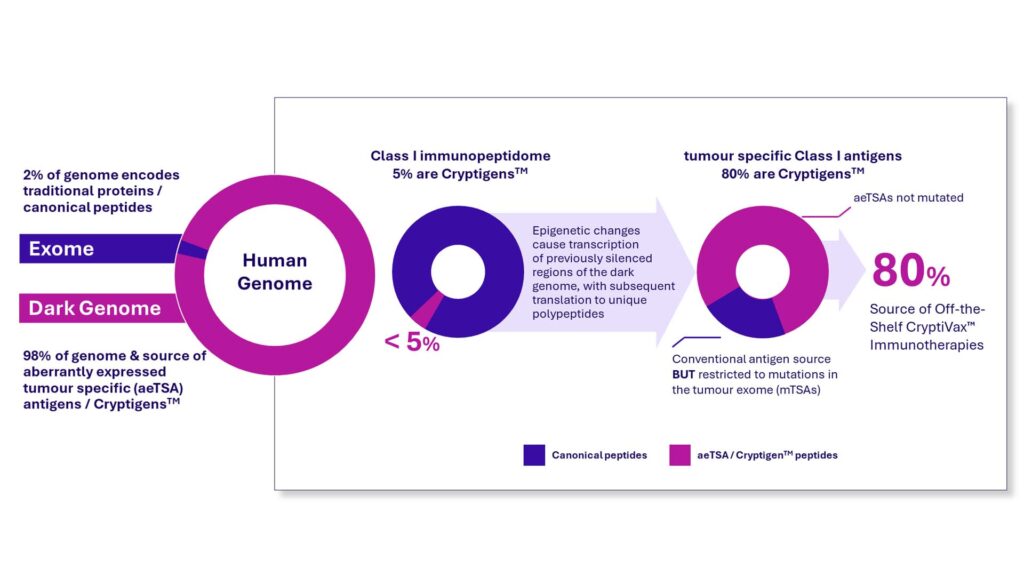
“We believe these proteins represent therapeutic targets. Precision therapeutics has traditionally explored only two percent of the genome. The remaining transcribed genome represents a vast, untapped opportunity,” said Alan Rigby, PhD, CEO at Epitopea. “Advances in deep, empirical mass spectrometry now permit a deeper interrogation of the MHC presented repertoire critical for immune recognition.”
Importantly, transcripts expressed in thymic medullary epithelial cells, which train the immune system to tolerate self, and/or determined to be expressed in the Adult Genotype-Tissue Expression (GTEx) dataset, are subtracted. aeTSAs derived from this expanded genomic space are highly cancer-specific, shared across patient subsets diagnosed with the same tumor type, and more abundant and immunogenic than previously discovered conventional neoantigens.
Epitopea’s vaccine platform incorporates iterative optimization principles borrowed from traditional hit-to-lead, lead optimization, and development candidate validation metrics leveraged in many small-molecule discovery campaigns. “We optimize not just the antigenic peptide but also other aspects of the construct that influence how these aeTSAs are presented to the immune system,” explained Rigby. This enhances T cell receptor diversity and promotes memory T-cell formation, critical for durability of response, which, ultimately, translates into meaningful clinical responses.
Epitopea’s first clinical program will be a Phase I, first-in-human trial in homologous recombination-proficient (HRP) high-grade serous ovarian cancer, beginning in 2026 in the U.K. HRP patients represent about 50% of ovarian cancer cases that do not benefit from PARP inhibitors. “In HRP ovarian cancer, patients currently have few precision-based options. We aim to change that in a first-line maintenance setting,” said Rigby.
The off-the-shelf RNA-based immunotherapies are built by adapting and optimizing the beads-on-a-string approach similar to those deployed in personalized cancer vaccines. The number of aeTSAs and the degree of sharing within a tumor indication suggest that, for many indications, a cancer therapy comprised of 25 aeTSAs would support a predicted tumor-specific median patient presentation.
…
Scientists remain determined to turn cancer, even the most difficult to treat cases, into a curable disease as the technologies highlighted in this article demonstrate. These innovative approaches, and others in development, have the potential to positively impact millions of lives. Only time and continued clinical trials will reveal the outcomes.